A wikipedia of Dr. D'Adamo's research
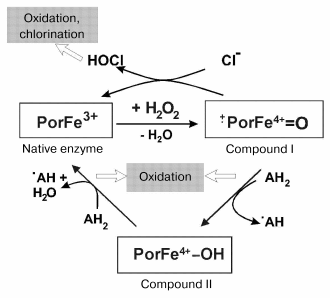
Difference (from prior minor revision) Changed: 8c8 < ====Functions f Myeloperoxidase==== to > ====Functions of Myeloperoxidase==== Immunology? C O N T E N T SDescriptionMyeloperoxidase is a lysosomal enzyme that is found in white blood cells, neutrophils. Myeloperoxidase is an enzyme that uses hydrogen peroxidase to convert chloride to hypochlorous acid. The produced hypochlorous acid reacts with and destroys bacteria. In many inflammatory pathologies, such as cystic fibrosis and rheumatoid arthritis, neutrophils are also causing tissue damage. Myeloperoxidase is also produced when arteries are inflamed and have rupture-prone fatty deposits. An inflammation in the arteries can lead to a blood clot and eventually to a heart attack or stroke. Myeloperoxidase is thought to be the most promising cardiac marker at the moment. In addition to that Myeloperoxidase is a good inflammatory biomarker for autoimmune, inflammatory diseases and cancer. Other cardiac markers, like troponin, myoglobin, creatine kinase isoenzyme MB and C-reactive protein, to which tests already exist, become present in the blood only after the damage to the heart has already been done. By measuring the myeloperoxidase level in blood it is possible to predict whether a person is in risk of heart attack or death in the following six months. Quoted TextFunctions of Myeloperoxidase
Discussion
MPO Defficiency in CandidiasisThe neutrophils and monocytes of most patients with deseminated candidiasis have been found to lack detectable levels of the enzyme myeloperoxidase (MPO). This rarely affects phagocytosis of the organism, but markedly decreases intracellular destruction, resulting in the organism persisting as intracellular inclusion. This perhaps affords an explanation to the persistance of some health care practitioners in employing questionable superoxide therapies in candidiasis. MPO synthesis is dependent on adequate tissue levels of both iodine and ascorbate, the therapeutic employment of which offers a more sound and safe approach. Links |
COMPLETE BLOOD TYPE ENCYCLOPEDIA
The Complete Blood Type Encyclopedia is the essential desk reference for Dr. D'Adamo's work. This is the first book to draw on the thousands of medical studies proving the connection between blood type and disease. Click to learn more
Click the Play button to hear to Dr. Peter J. D'Adamo discuss .
|
The statements made on our websites have not been evaluated by the FDA (U.S. Food & Drug Administration).
Our products and services are not intended to diagnose, cure or prevent any disease. If a condition persists, please contact your physician.
Copyright © 2015-2023, Hoop-A-Joop, LLC, Inc. All Rights Reserved. Log In